
Over the years that I have been working, starting as a junior C&Q engineer, there has been an increasingly blurred line between Commissioning and Qualification that at times, it is difficult for my counterparts in the construction management team (CMT) to exactly understand the division (or integration) of tasks between the CMT and the CQV teams. This is largely due to the move of the industry from a traditional V-Model into a hybrid approach, and more recently, more accepting towards a full risk-based approach CQV model.
In this article, I would like to share with you my take on the top five myths about Commissioning and Qualification (CQ) in the pharmaceutical industry and debunking them in the process!
Please read my previous article on the IQ, OQ, and PQ in the pharmaceutical industry where I have tried using the -20°C freezer, as an illustration, to better explain the concept.
1. All Facilities, Utilities and Equipment (FUE) need to be Commissioned AND Qualified.
NOT TRUE! With increasing cost awareness of manufacturing companies and the push for greater product and process understanding from the regulatory agencies, the limited resources need to be funnelled to the FUEs that need them. For e.g. one would not place the same level of testing rigour onto a -20°C freezer that is used in a general industry compared to the same -20°C freezer used to store temperature-sensitive GMP raw materials.
The starting point is normally the System Impact Assessment (SIA). Following the ISPE Baseline Guide Volume 5 – Commissioning & Qualification Volume 2, a FUE is categorized as a direct or indirect system after methodologically answering 8 guided questions. The outcome is normally documented to form part of the C&Q Execution Plan.
The backbone of a successful qualification programme is a robust commissioning effort. All FUEs, regardless of direct or indirect, shall be robustly commissioned with Good Engineering Practices (GEP) commensurating with the perceived risks to the product and patients. For those of you that have been in the industry, you would have known by now that a poorly commissioning system would not pass qualification, and that’s a given.
2. Commissioning activities (direct systems) are managed by CMT and will be handed over to the CQV team with all issues solved!
NOT TRUE! Gone are the days when there was a clear demarcation between the different phases in a project. Current day projects emphasize precision and accuracy while optimising the overall project delivery timeline.
Shifting away from the traditional approach (see diagram), the best practice integrated approach sees the “Qualification” team onboarded earlier in a project life-cycle – as early as during the engineering phase. The “Qualification” team will work alongside the Engineering team ensuring that the User Requirement Specifications (URS) are fulfilled in the design through Design Qualification (DQ) process before the equipment is purchased and/or the design finalized.
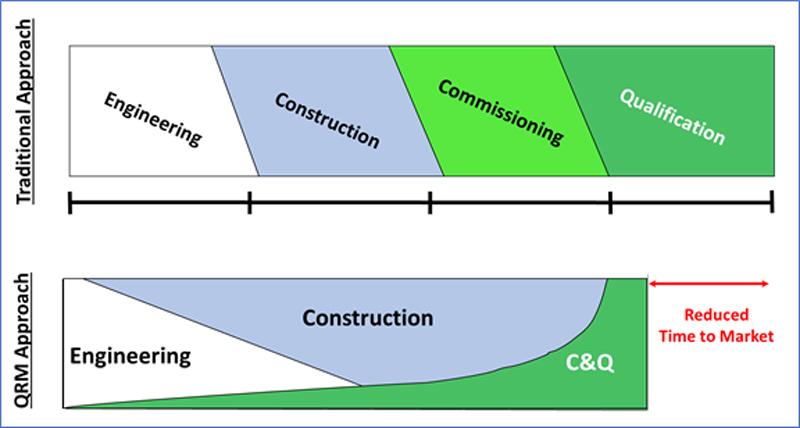
Given the right opportunity, the Qualification team works alongside the CMT Commissioning Team in an integrated fashion, identifying the opportunities to test the equipment at the earliest availability while still ensuring that the tests performed are valid and could be used as part of qualification.
Tying into the earlier article on -20°C freezer qualification processes, the C&Q of the -20°C freezer could start as soon as the -20°C freezer is delivered to the site through CQ Out of Place (OoP) strategy with remaining activities completed when the -20°C freezer is finally moved to location. This approach might not work for all types of equipment and the risk versus benefit assessment shall be performed with the CMT, Subject Matter Expert (SME) and Quality Assurance (QA) in general.
This comes to show that commissioning activities are not just managed by CMT in isolation. Early engagement with the Qualification team could save a massive amount of time in execution, and even detect problems early on the project.
3. Commissioning and Qualification are REPEATING activities but named differently.
YES and NO. To tackle this issue, we first need to revisit the traditional CQ V-Model versus the Risk-Based Approach Model.
 |
 |
The figure on the left is extracted from the ISPE Baseline Guide Volume 5 – Commissioning & Qualification First Edition while the right is from ISPE Baseline Guide Volume 5 – Commissioning & Qualification Second Edition.
Given that the ISPE Baseline Guide Volume 5 – Commissioning & Qualification First Edition has been widely used and accepted since its publication in 2001 and has only been replaced by the Second Edition in 2019, the supposed “legacy” reaction would generally be, “Oh, all the tests in the Commissioning Protocol will be repeated in the subsequent IQ, OQ and PQ”. And to that part, the answer “YES” applies that indeed that was the practice for many years in between the introduction of the risk-based approach.
Fast-forwarding into ISPE Baseline Guide Volume 5 – Commissioning & Qualification Second Edition, the emphasis as I could relate to and communicate with you is – Not to get hung up on terminologies. The required tests to prove SYSTEM is “fit for intended use” triumphs.
As shown in the diagram extracted from the ISPE Baseline Guide Volume 5 – Commissioning & Qualification Second Edition, the entire model is more holistic. The manufacturing company will need to have more process and product understanding in identifying the critical design elements that need to be challenged and tested. How and when these challenges/tests are to be executed is entirely left to the manufacturing company with the strategy and justification clearly documented in the CQ Execution Plan, typically approved by Quality Assurance. If planned, coordinated, and executed religiously, having the mindset of “End in Mind”, Commissioning and Qualification ARE NOT repeating activities, but complementary.
Using the -20°C freezer with Operational Qualification (OQ) activity of empty chamber temperature mapping activity, the test performed in CQ Out of Place (OoP) is admissible as part of the qualification package as a risk-based approach based on the equipment type. The detection of any lack of performance would be captured during loaded chamber temperature mapping at PQ.
4. Well, if commissioning and qualification are the same (or almost the same), let us drop commissioning and dive straight into qualification to save time in the project.
IT DEPENDS. Let us assume that we are adopting a risk-based approach for the Commissioning and Qualification following the guidance of ASTM 2500–13. One of the key concepts is “Use of Vendor Documentation” – including vendor test documents that may be used as part of the verification documentation, provided that the manufacturing company has assessed the vendor as a part of the vendor qualification programme.
One of the downfalls of the risk-based approach is not planning sufficient execution duration for equipment because of the assumption that no repetition of activities is required because the vendor is “impeccable” in their execution. This assumption cannot and shall not be made if there is no thorough assessment of the vendor capability and track record in delivering the scope.
Coming back to the example of -20°C freezer – the level of confidence that is accorded if the -20°C freezer is manufactured by an equipment manufacturer that has over 50 years of experience with international presence will be significantly different from that of a new equipment manufacturer that has just entered the market. Having said that, ultimately, it boils down to the rigour of the vendor qualification to ascertain if a shakedown phase is necessary before jumping into qualification. If indeed a shakedown is required, perhaps a commissioning phase before the qualification might be justified. This is especially true for more complicated systems.
5. Good Documentation Practices (GdP) do not apply to the commissioning documents.
NOT TRUE! GdP should be applied to all documents regardless of whether they are commissioning documents or qualification documents. GdP following the Data Integrity (DI) principles should be upheld at all stages of the projects. This is especially true if commissioning activities/documentations are integrated as part of the qualification activities. During an audit, the inspector could access all areas of documentation for a system. Therefore, they should meet the GdP requirements.
At No deviation, we partner with you to help you with executing your CQV process, troubleshooting or your CAPA, or implementing new digital solutions. Reach out to us for a full risk-based paperless validation with integrated commissioning and test plan or to bring efficiency to your existing paper-based IQ, OQ execution. You can also visit our website for more information on our services.



