As the pharma industry evolves, digital transformation is slowly gaining importance. Digital transformation can affect many parts of the lifecycle of a drug. However, it is not a smooth journey as it presents its own set of challenges as well. Read on to find out more about digital transformation and Quality 4.0 in the pharma industry.
During the ISPE Singapore Affiliate Conference & Exhibition 2022, Santhosh Francis (Area Vice President – Sparta Systems, a Honeywell Company) spoke about Quality 4.0 and how to accelerate it through connected life sciences. For those who are not familiar with Quality 4.0, Quality 4.0 can be understood as a term which defines the future of quality and organizational excellence within the context of Industry 4.0 through the use of digital and disruptive technology to improve the quality of an organization with the help of quality professionals. This is done to achieve the aim of improving the quality of an organization and its ability to provide customers with high-quality products in a reliable manner.
Santhosh shared that under the 4.0 framework, it is paramount that data and information in a connected environment and infrastructure be processed in a manner which translates to improved efficiencies, cost reduction, as well as improved standards for the company. However, the situation in life sciences presents its own set of differences, as patient safety and security are of utmost importance and there is always the consideration of regulatory and compliance during decision-making processes. In addition, with data collection and processing using Artificial Intelligence (AI) and Machine Learning (ML), there are also security-related challenges.
So why is there a need to focus on quality? There are 2 aspects in this regard:
- Cost of quality: The cost that is required to maintain and manage quality, as well as the additional costs which are incurred as a result of poor quality.
- Patient: Being the heart of everything that is being done in the life sciences industry, it is important that a safe and efficacious solution be delivered to the patient. Despite supply chain challenges, we still have to ensure that medicines can reach patients promptly.
Figure 1: Some Quality 4.0 use cases
The Quality 4.0 framework aims to achieve interconnectedness between people, processes, systems and technologies, to ensure that the data obtained allows for the right decisions to be made in a timely manner.
However, there are some challenges that could slow down a company while moving towards achieving Quality 4.0, and these can be attributed mainly to 3 factors:
- Perception of quality: There are times when the focus on quality and compliance results in disconnected systems and this results in undesirable outcomes for the organization.
- Data deserts: Data are in silos or islands. To achieve an effective working system, it must be ensured that the different applications across an enterprise can be integrated and work well with each other.
- Supply chain complexity: Around 60% of the data lies outside the organization. Data and elements within the external supply chain have to be extracted and properly integrated to allow for effective decision-making.
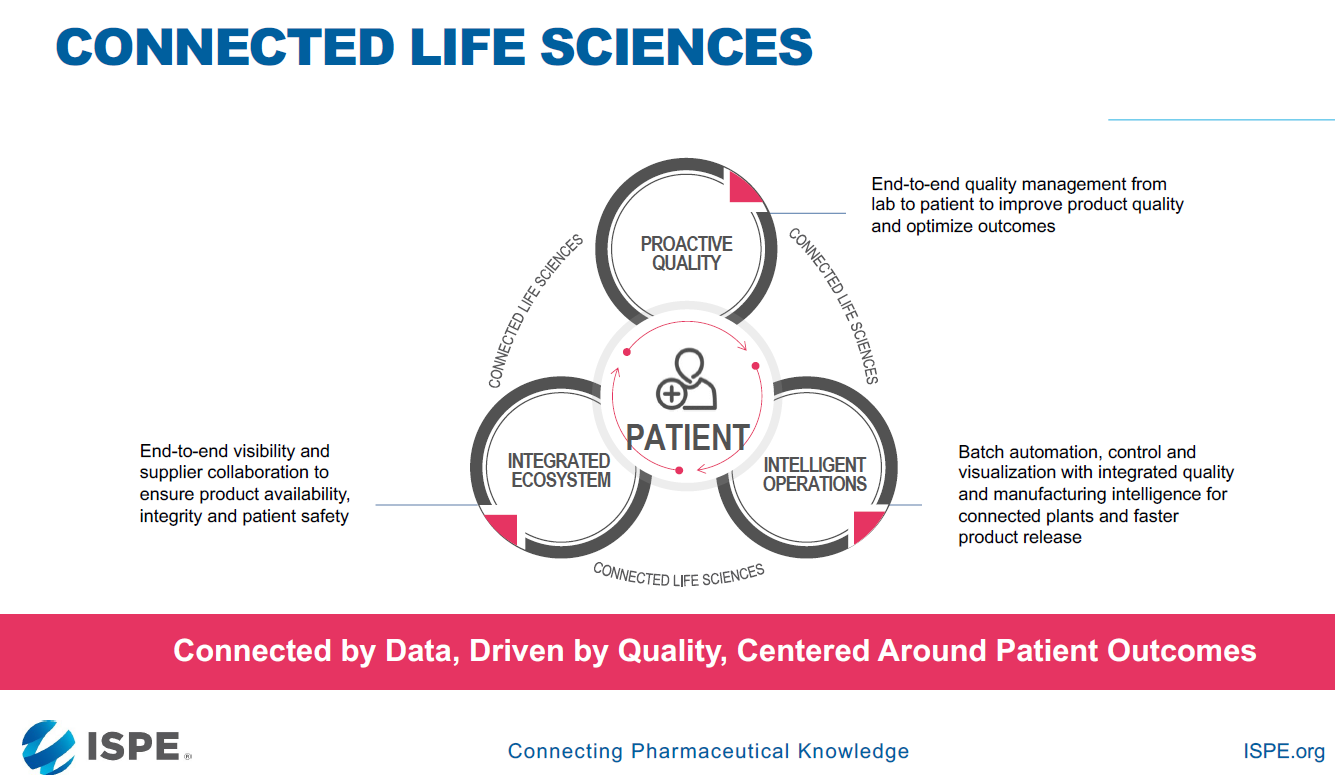
To deal with the challenges listed above, the concept of connected life sciences is introduced to help organizations in their journey towards Quality 4.0. In this proactive approach to quality, data is analyzed proactively to prevent issues from occurring in the future and for optimized outcomes in product quality. This can be achieved with the help of intelligent operations where batch automation is interconnected with manufacturing intelligence for faster product release. Lastly, it aims to achieve an integrated ecosystem, where there is end-to-end visibility and supplier collaboration to ensure product availability, integrity and patient safety.
Figure 2: Connected life sciences and its components
Finally, in the journey of moving towards Quality 4.0, there are a few important pointers to note in minimizing the risks:
- Get your data and your organization ready: What is the organization’s culture and attitude towards achieving Quality 4.0? What is the amount of data that is to be dealt with and its availability?
- Focus on outcomes over technology: Do not go digital for the sake of going digital. Look at the outcomes and benefits that can be derived for both the organization and the patient.
- Go slow before you go fast: Start small and then scale up.
In our next article, we will discuss a few factors leading to this shift towards digitalization and the challenges in implementing digital solutions in the pharmaceutical and biopharma space. Stay tuned!
At No deviation, we have extensive experience in executing and implementing digitalization projects in the form of the Kneat paperless validation software, the PlantQuest critical asset management software, and the Mirrhia Environmental Monitoring System (EMS) & Contamination Control software for pharmaceutical and biopharmaceutical companies. If you have any questions regarding such digital solutions, please contact us at hello@nodeviation.com.
You can also visit our website for more information on our services.