Suspension of a site’s GMP certification imposes substantial stress and impact on business, as well as the potential shortage of market supply.
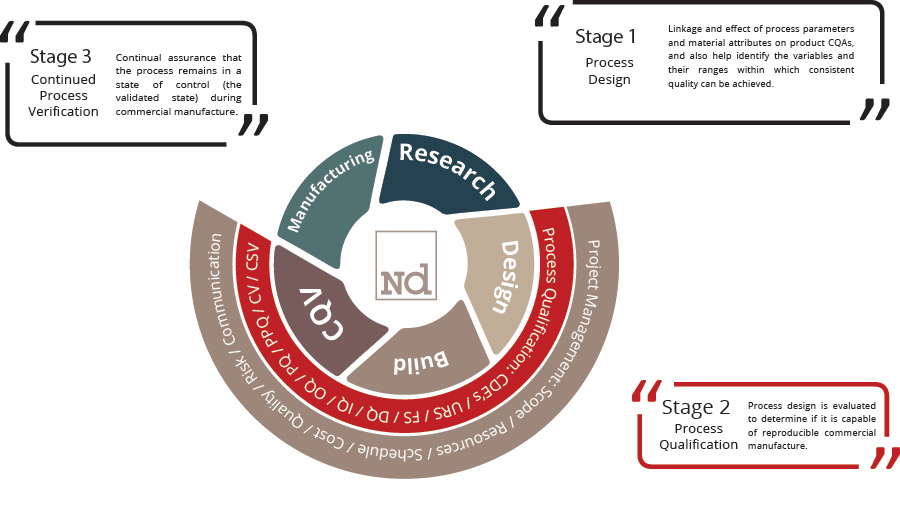
This case study outlines how the No deviation team supported a China-based biologics manufacturer to bridge the compliance gaps and achieve the ‘State of Control’ – a condition in which the set of controls consistently provides assurance of continued process performance and product quality. (ICH Q10)
Project background
Manufacturing Site Brief:
Product: Injectable – Sterile
Facility: Biologics Drug Substance + Aseptic Fill Finish
Major Gaps to Compliance:
No deviation was requested to perform a site-wide gap assessment based on which the execution strategy was established.
- Water System: Due to poor system design, construction and maintenance as well as inadequate OOS investigation, there was a lack of assurance of PW and WFI quality.
- HVAC System: Similarly, the HVAC system was observed as having the same issues hence the cleanroom condition was out of control.
- Process Equipment: Due to poor design and construction, the manufacturing process required intensive manual intervention, which was not instructed by or incorporated in batch documents.
- Qualification and Validation: Particularly insufficient qualification and validation, i.e. lack of evidence of cleaning effectiveness.
- Fundamentally, a lack of Quality Mindset.
Project strategy
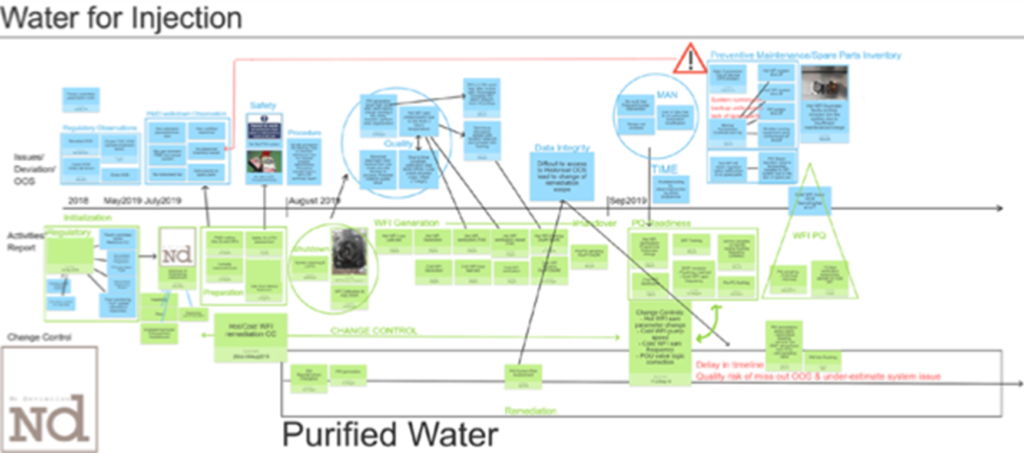
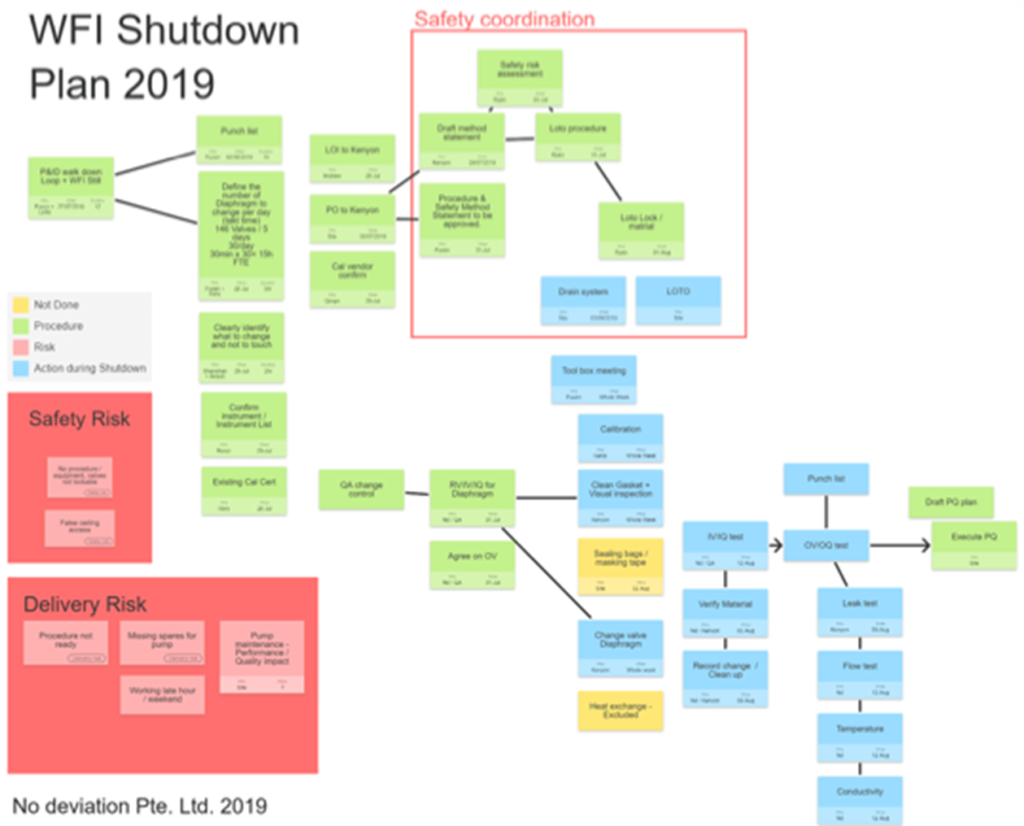
For complex topics, various tools were utilised to facilitate the multidisciplinary team discussion to understand the true root cause, risks and opportunities. An illustration of planning a water system remediation strategy is given below –
No deviation’s service scope
- Project Management: The No deviation project manager established various PM tools including Project Charter, Project Scheduling, Risk Register, Change Management, Tier Meetings and Project Reporting. Various stakeholders including suppliers were managed under the No deviation team.
- Process Equipment Remediation & Qualification: Equipment was assessed for the required manufacturing process and quality attributes. Remediation was conducted and the system qualified for the impacted functionality.
- Water System Remediation & Qualification: System improvement was done ranging from mechanical installation, automation control, maintenance, sanitisation programme and monitoring programme.
- HVAC System Remediation & Qualification: System issue was identified from benchmarking, based on the results various remediations (i.e. filter changeout, duct re-routing), tuning, balancing were conducted prior to qualification.
- Calibration Management: A calibration plan and methods were established, critical instruments were calibrated.
- Filling Line Assessment & Qualification: Filling line (with open RABS) was assessed for its capacity and sterility assurance.
- Maintenance Programme Establishment: Programme was established based on FMEA assessment.
- Inspection Support: Nd Compliance SME supported NMPA inspection from readiness to CAPA establishment – GMP Certification Achieved.
Major Achievement
The short term and most urgent objective for the project was to re-gain the GMP certification. During the duration of the whole project, the No deviation team had been working intimately with the client’s team and other suppliers. Throughout this memorable journey, our substantial achievement was in building a quality culture which helped shift the mindset from passively following instructions to taking ownership of quality, and ultimately, to proactively identifying and mitigating the quality risks.
With such a quality culture, we’ve also identified long term objectives including the development of fit-for-purpose training, alignment of quality and HSE standards to a global level, management of internal and external inspections and alike.
We hope this case study gives you an insight into how we support our clients in regaining their GMP certification and help build a quality culture in the organisation.
At No deviation, we partner with you to help you build the quality in your product. Reach out to us to know how we can help you achieve, retain, and regain cGMP compliance.
You can also visit our website for more information on our services.