So…you have your Kneat Instance validated and you are eager to roll it out to all areas of your organization. Hold your horses and allow me to share with you the key considerations before scaling up and out, on the use of the world’s most advanced validation technology, KneatGx.
 Why the need for Kneat Governance?
Why the need for Kneat Governance?
The fundamental driver of Kneat implementation, in my opinion, is to create a consistent approach through the use of scalable and controlled templates. If this can be achieved, efficiency and effectiveness will be realised. However, one cannot leave it to chance for that to be realised. Well thought-out and conscious efforts are required – in comes GOVERNANCE. The governance should need to be driven from the centre while engaging and gaining feedback from the satellites sites/departments to allow for the PDCA cycle.
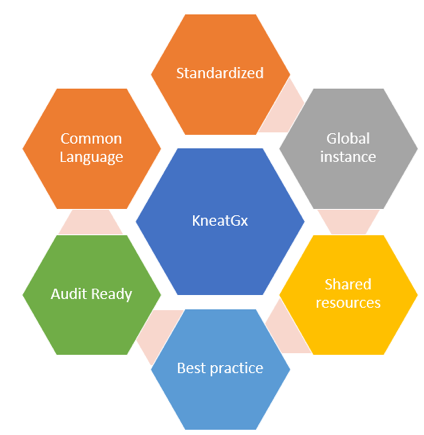
With KneatGx sitting at the centre of the diagram, the elements surrounding it would need to be subjected to the inspection eyes of governance. In this article, we will not be detailing every single element, but I would like to pick up the elements of “Standardized” and “Shared Resources” for discussion purposes.
On the element of “Standardized” and recapitulating on the notion of the fundamental driver for KneatGx implementation, KneatGx starts with identifying the process through a process mapping exercise with the output of a defined Discipline Map including the associated Document Types. For e.g., if equipment Commissioning & Qualification (CQ) were identified as the process of interest, the resulting Discipline Map would be named “Equipment Commissioning and Qualification”. Associated with the “Equipment Commissioning & Qualification” Discipline Map could potentially include the following Document Type: System Level Impact Assessment (SLIA), Quality Risk Assessment (QRA), User Requirement Specification (URS), Design Qualification (DQ), Design Review (DR), Commissioning & Qualification Test Plans etc. – and with each of the Document Type, an associated Document Template, and the Approval Template. It is apparent that the repeated use of the word “template” points towards an important aspect of KneatGx.

As the usage of KneatGx starts to increase, it is natural that there would be increasing requests for different Discipline Maps, Document Types, Document Templates, Approval Templates, etc. It is imperative that governance is exercised over the administration of the templates including the responsible group of users administering them. One could imagine if each request were fulfilled, very fast and very soon, the desired approach will diverge and the KneatGx Instance will be riddled with site/department-specific wants which may or may not be aligned with global requirements.
Second, on “Share Resources”. With a robust platform such as KneatGx and the proper management of Subject Matter Experts (SME) within the organization, a high degree of economies of scale and economies of skills can be realized to raise productivity for all sites. The key word is “proper management”. How could we collectively and systematically ensure that the knowledge from ONE global SME can effectively reach out to MANY other local sites? Using a controlled accessed centralized library, where test scripts are created and approved by a select few that are qualified, allows the sharing of resources across the site with the latest/greatest version of those tests. It’s apparent, therefore, that good governance is required to manage tests and the personnel that has access to this workspace.
There you go! A little flavour on the importance of governance when rolling out the KneatGx Instance within the organization.
If any of the points strike a chord with you, please reach out to hello@nodeviation.com for a more in-depth discussion on the importance of governance including the challenges that you might face.
You can also visit our website for more information on our services.



