Our client’s email mentioned:
“As we are considering using Kneat as a digital tool for Validation activities within the project, we would like to organize a meeting with No Deviation who is already involved in the C&Q Initiative Kneat Deployment. The aims of this session are:
- To explore the benefits of using Kneat
- To discuss the best practices of Kneat implementation
- To explore the support that No deviation could provide…”
There are a couple of questions to be answered before diving into replying to the email requests and that includes the following:
- What are validation activities?
- How are these validation activities perceived to be different from that of qualification?
- What are the features of Kneat that could be tapped into, to support the request?
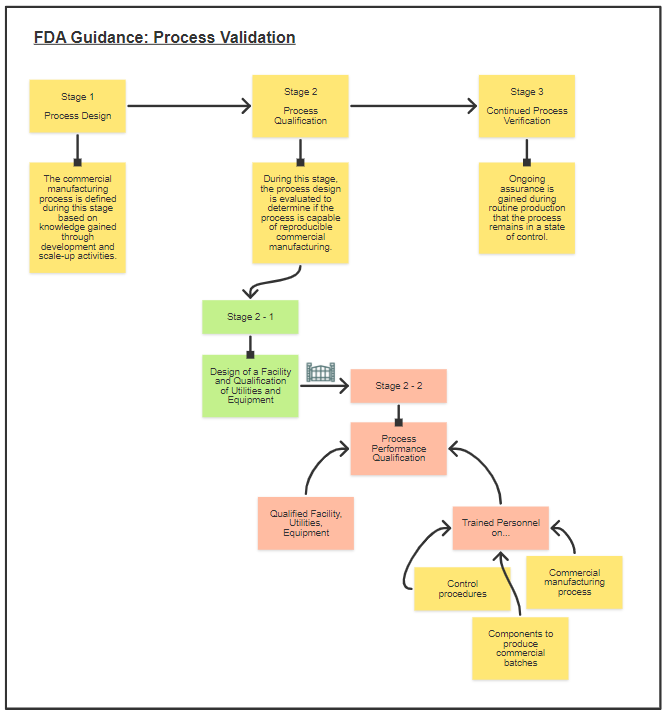
To answer points 1 & 2, one would need to know the applicable regulatory definition. In this context with the querying client, the FDA publication “Guidance for Industry Process Validation: General Principles and Practices” shall be used.
Extracting the definition from the FDA Guidance, “Process validation is defined as the collection and evaluation of data, from the process design stage through commercial production, which establishes scientific evidence that a process is capable of consistently delivering a quality product. Process validation involves a series of activities taking place over the lifecycle of the product and process”.
The illustration below is my quick draw-up of Process Validation.
As one can appreciate, the notion of qualification as a standalone process is not exactly true. It is an integral part of process validation. Referring to the FDA guidance, Stage 2: Process Qualification is a 2-prong process, with the first being the Facility, Equipment and Utilities qualification process to ensure that these “hardware” are fit for their intended use. The second prong would then be ensuring that the personnel/environment/SOPs coupled with the qualified Facility, Equipment and Utilities results in consistently producing APIs and drug products meeting those attributes relating to identity, strength, quality, purity, and potency.
On to point 3 – What are the features of Kneat that could be tapped into, to support the process validation? The simple answer would be ALL of the features. Assuming that a robust Process Validation programme has been put in place, KneatGx as an execution tool could bring consistency, accuracy and efficiency. These are the few powerful features in KneatGx that will bring benefit to the programme:
- (A)All in one platform – All related documentation at different stages of Process Validation can be captured in KneatGx, which is a 21 CFR Part 11 compliant platform. Centralization of documentation and availability at the click of a button translates to time-saving in whichever activities you are doing.
- (B)Cross-referencing made easy – With the intimate relationship from one stage of Process Validation to another, the ease of cross-referencing allows for the quick pick-up of crucial information to support execution and audits.
- (C)Sharing of information – Through the use of Templates, smart ID Tag, system properties and library properties, repeating information such as equipment tags number, the outcome of an assessment, acceptance criteria etc. can be used and reused in a multitude of documents ensuring consistency.
- (D)Audit readiness – With all the information captured in a 21 CFR Part 11 compliant platform, all documents inherently meet the Good Documentation Practices (GDP) and ALCOA+ principles by design. On top of that, the required documentation to support an audit is available within a click away through the clever implementation of cross-referencing.
With No deviation’s technical expertise in supporting the development of a process validation programme and our expert use of KneatGx, we had managed to respond to each of the queries and concerns from the client. I’m pleased to share that the meeting went well and we are now working hand-in-hand with the client, progressing to the next step in their process validation journey.
Get in touch with us at hello@nodeviation.com for a non-obligatory discussion on how we can help you discover the full potential of the world’s most advanced validation technology, KneatGx.
You can also visit our website for more information on our services.