In our previous posts, we explained why there is no better time than now to work in the pharmaceutical industry. It is especially true for the year to come, if you live in Singapore or plan to relocate here. With the current COVID-19 situation, much more manufacturing power is needed, and Singapore is a unique hub for this. With its stable geopolitical situation, educated workforce and well-managed Covid situation, Singapore has minimised project, and manufacturing disruption.
Do you want to reorient your career? Or, are you fresh from school and want to embrace a future? No deviation is specialised in the commissioning, qualification and validation activities for the pharma and biopharma industry, so here are a few points for your consideration.
Like in any profession, there are a series of attributes you will need to excel in your work. I once saw a definition for ‘capability’ that I would like to summarise in the image below:
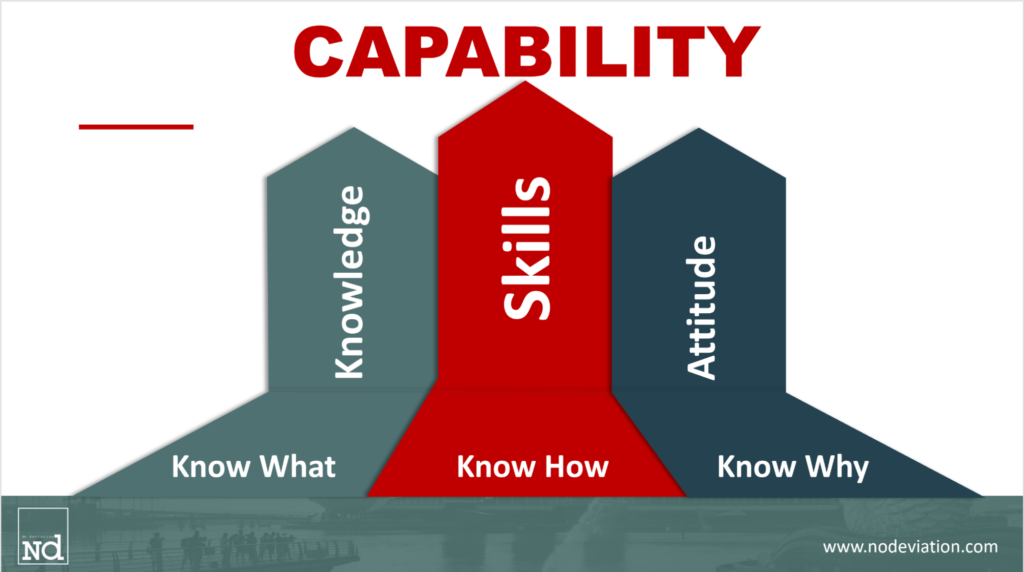
Based on the above image, let’s look at the specific knowledge, skills, and attitude that will make you a capable CQV engineer.
Know Why:
First of all, you need to know why you are doing equipment testing and process validation. Besides being a regulatory requirement (refer to the EudraLex Volume 4 Annex 15 for some guidance), you will do this so that patients will receive a quality product on time, every time. This means, you need empathy.
Empathy for the patient whose well-being depends on your job, empathy for the operator that will use the equipment, empathy for the engineer that will maintain the equipment, and empathy for the quality department that will need to trust the performance outcome of the equipment.
Know What:
You will also need to know what to test, what is essential, and where to focus. To understand what to test, we refer to what could affect the product quality, also called critical quality attributes (CQA), as defined in the Q8(R2) Guideline, e.g. sterility, potency, purity,…
Processes and equipment are designed to control a series of parameters. Some are functional; some are critical process parameters (CPP) e.g. temperature, time, pressure, mixing speed, heating rate,…) as they help build the quality attribute into the product. In the commissioning phase, you will make sure the equipment is functional, and you need to have a deep understanding of the equipment. In the qualification exercise, you will demonstrate that the system is fit for its intended use, and you will need to have a good understanding of the process/intended use. The intended use is using a system in the context of your manufacturing, for your product, to support your patient needs.
To identify what is critical, we use a quality risk assessment as part of the quality risk management defined in the ICHQ9 (QUALITY RISK MANAGEMENT), where you will ask yourself: What might go wrong? What is the likelihood (probability) it will go wrong? What are the consequences (severity)? From there, take the appropriate level of effort to mitigate unacceptable risk and demonstrate your mitigation is effective (Testing).
Know-How:
Developing the skills to test a system is something you will need to learn by experience. There are no CQ schools today, and training is scarce. There is no better school than the start of facilities. The skill of focusing on what is important and writing a test script that will give the assurance the system works as per intended use, and the skill of identifying any risk of failure, require theoretical and field experience.
The good news is that you can apply previous experience to the job if you ever troubleshoot something, understand how things work, project yourself on the desired state, and have a process background and basic physical law understanding. Commissioning and qualification should not focus on the testing part only. They should focus on making sure that the test, equipment, system, and process will work and be robust.
The following graphic explains the different skills that can help you become a CQV engineer or enhance your current capability to be one.

At No deviation, we partner with clients to help them with executing their CQV process, troubleshooting or their CAPA, or implementing new digital solutions. Reach out to us if you are interested in knowing more or would like to apply for a position with us.
You can also visit our website for more information about No deviation and its services.


