Resources & Inspiration

Importance of governance in the implementation of Kneat’s paperless validation solution for the pharma industry
So…you have your Kneat Instance validated and you are eager to roll it out to all areas of your organization. Hold your horses and allow

The importance of a Project Charter in pharma project management in Europe
“Begin with the end in mind…” When it comes to Project Management, it is imperative that we know ‘why’ from the start and this, among

Key considerations around environmental monitoring in pharma manufacturing
Clean rooms are at the heart of the manufacturing of pharmaceuticals, medical devices, in-vitro diagnostic solutions and electronics. Each manufacturer makes a commitment to the

Talk to the expert: Morgan O’Brien, General Manager Europe, No deviation
1. Tell us more about your role at No deviation. At the moment, I am heading our Europe office back in my hometown of Cork

5 things to know about Mirrhia – an environmental monitoring system for the pharma industry
Patient safety is always the top priority in the pharmaceutical industry. Mirrhia, an environmental monitoring system and contamination control ensures patient safety and faster production

Paperless Validation training for the pharmaceutical industry with No deviation
Further to my earlier post on “No deviation and Paperless Validation: how it all began”, I wrote that No deviation has Kneat Academy certified Master
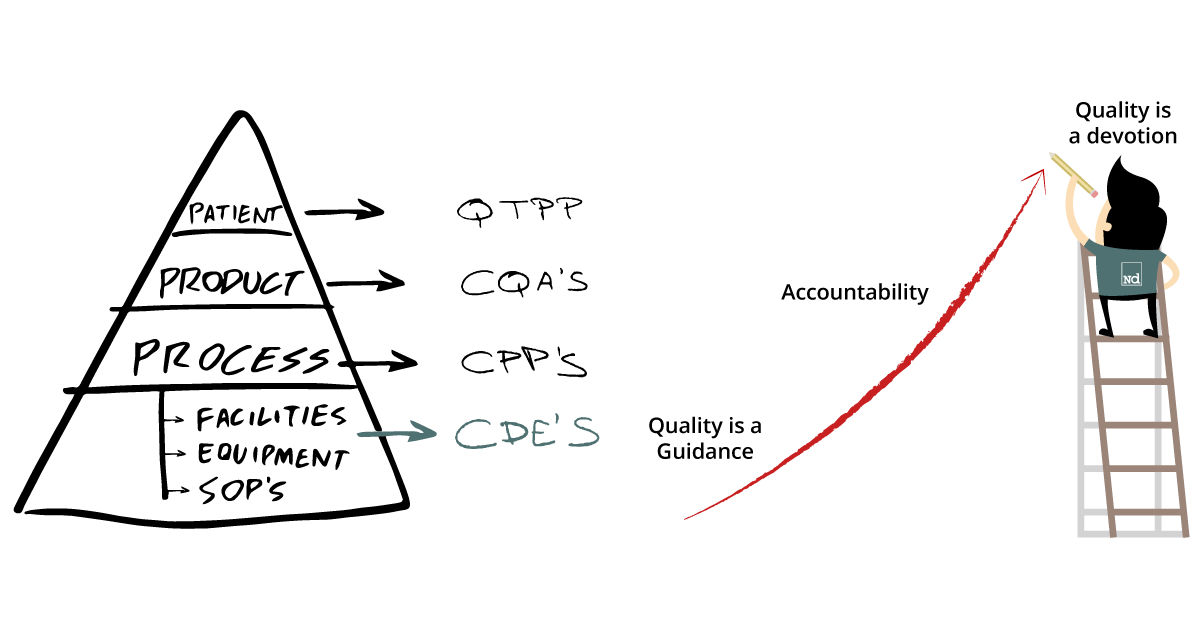
How to reach a quality mindset in the pharma industry
In the pharmaceutical industry, quality is one of the foremost considerations. Technological evolution has enabled new ways of delivering quality, which go beyond compliance. In

Talk to the expert: JoonLeong Ng, Director, No deviation
1. Tell us more about your role at No deviation. It would be a very long story if I had to detail all my activities

